Patients

The Zenfuze System is a minimally invasive solution for patients suffering from low back pain. Following conservative treatment methods, Zenfuze may be an option. The Zenfuze System can treat conditions such as spinal stenosis, spinal instability, and degeneration. Zenfuze may provide patients with a permanent solution for the conditions associated with low back pain. The majority of patients will notice immediate improvement to many or all of their symptoms.
No Metal
The Zenfuze Implants are derived from allograft bone.
Safe
The Zenfuze System was developed to prioritize patient safety.
Minimally Invasive
Studies have shown that patients undergoing MIS procedures report less pain and require smaller doses of pain relievers than patients undergoing traditional surgeries.
Excellent Outcomes
Physicians using the Zenfuze System have reported exceptional outcomes.
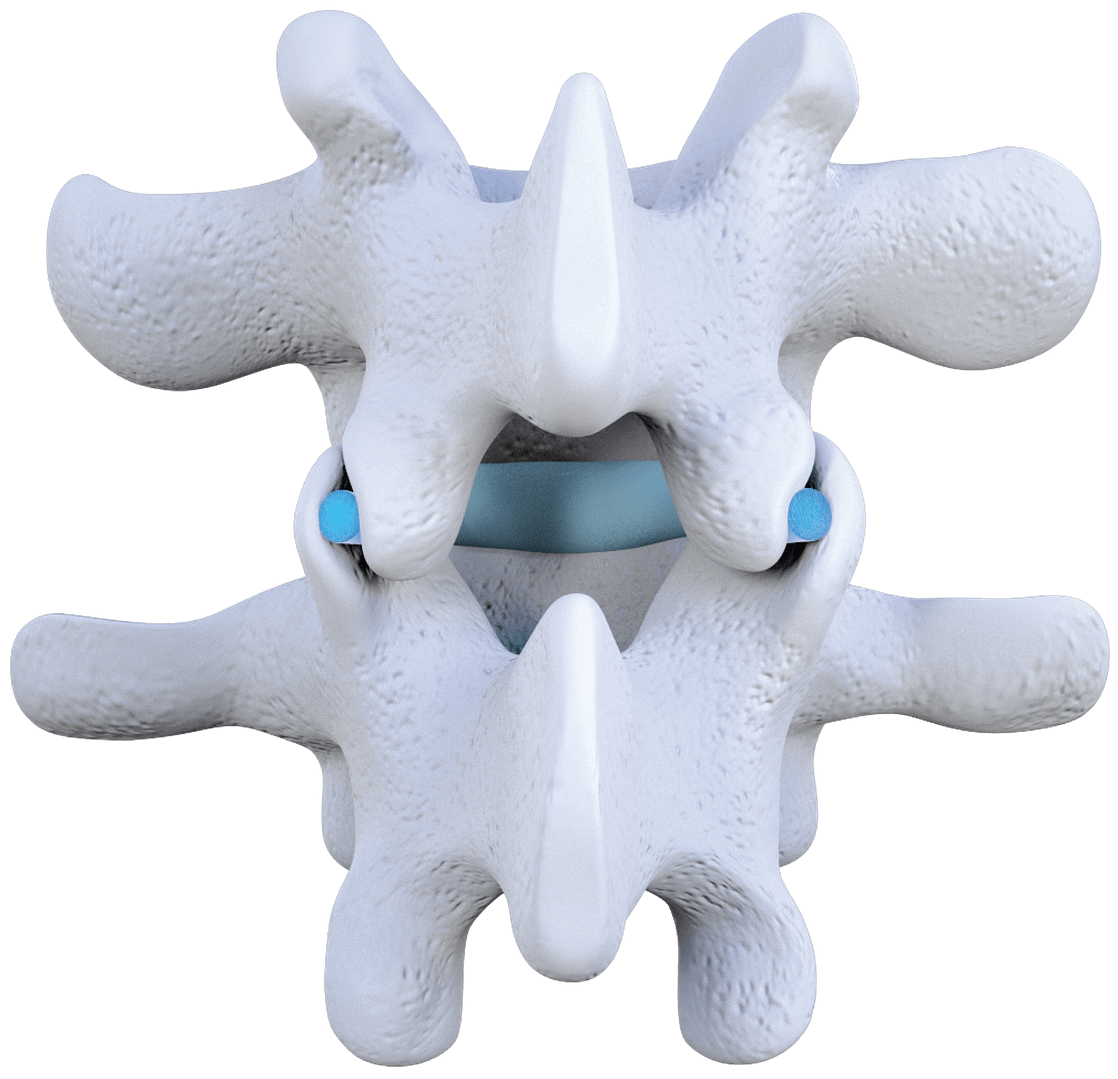
Zenfuze System
Allograft implants are placed into the facet joints to re-establish natural joint orientation and separate the joints to reduce inflammation. The Zenfuze System has been designed to stabilize the spinal segment by limiting motion of the joints. Zenfuze still allows for micromotion, which creates an optimal environment for fusion.


What Is A Minimally Invasive Procedure?
Minimally invasive surgeries cause less postoperative pain and discomfort. Studies have shown that patients undergoing MIS procedures report less pain and require smaller doses of pain relievers than patients undergoing traditional surgeries.
Conservative treatment
Conservative treatment methods of lumbar back pain can be used, but many are short-term answers to a long-term problem. Back pain sufferers often initially try clinical solutions such as physical therapy, pain medication, steroid injections or radiofrequency ablation. If the pain is still present following the conservative treatment methods, there may be an underlying cause to the lumbar back pain, such as spinal stenosis or instability. At this point, your physician may recommend the Zenfuze System.




Need Help?
Frequently Asked Questions
If you have tried at least 3 months of conservative, non-surgical, treatment methods (physical therapy, medications, injections) with only temporary pain relief, you may be a candidate for the Zenfuze System.
Conservative treatment methods may only offer temporary relief for pain. The Zenfuze System may be a permanent solution for low back pain.
Zenfuze implants are custom milled cortical allograft. Donated human tissue production and processing is regulated by the FDA under CFR Part 1271 – HUMAN CELLS, TISSUES, AND CELLULAR AND TISSUE BASED PRODUCTS. Zenfuze Allograft is processed by licensed tissue banks which are fully compliant with all FDA requirements for tissue processing in the United States.
Zentech Spine and its partners are compliant with regulatory organizations.
- Zentech is a registered tissue bank.
- Zentech is registered with the U.S. Food and Drug Administration (FDA).
- Instruments are manufactured by ISO 13485 certified companies.
- Allograft partners are accredited by AATB and FDA.
- Tumor
- Infection
- Gross Instability
According to our physicians, you may expect to return to Activities of Daily Living (ADLs) within 2 weeks. A back brace will be worn for 6-8 weeks following your procedure. Special attention should be paid to limiting lumbar spine flexion and extension motions and axial rotation. During this period, it is recommended that you not lift over 10 pounds and that you avoid the use of NSAIDs.
Each patient is unique. Specific recovery guidelines will be provided by your physician.
